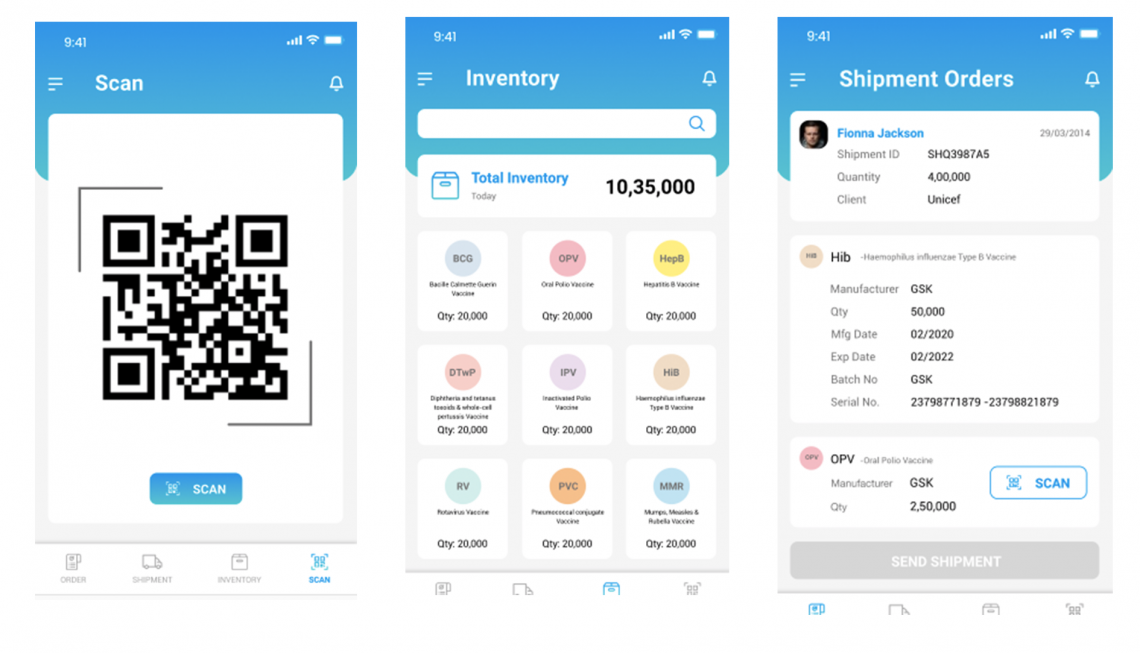
At StaTwig, our team has designed and developed two products: VaccineLedger and BabyBoo. With these two products StaTwig can track the journey of each vaccine from manufacturer to child.
At each touchpoint in the vaccine’s journey critical information about it such as temperature, humidity, chain of custody and location are recorded on a blockchain ledger. This immutable data is accessible by the stakeholders so that the products deliver trust and transparency in the supply chains. Every vaccine or shipment is tagged with a QR code. The QR code provides a unique identity for the product. This unique ID is used to store data against it. As the products change hands in the supply chains the stakeholders scan the QR code using the mobile based application.

Related Stories

AGUA: Revolutionizing Global Collaboration for Funding Transparency (with Growth Graduate Atix Labs)